Proven skin clearance
In 2 clinical trials, more than 5
out of 10 people saw at least 90% clearer skin at 4 months
(16 weeks)
Proven skin clearance


Proven skin clearance
In 2 clinical trials, more than 5
out of 10 people saw at least 90% clearer skin at 4 months
(16 weeks)
Clearance over time
Percentage of people on ICOTYDE who saw at least 90% clearer skin


2x as many people had completely clear skin
2x as many people had
completely clear skin
with ICOTYDE compared to another psoriasis pill
At 6 months (24 weeks), over 3 out of 10 people taking ICOTYDE had completely clear skin, compared to less than 2 out of 10 people taking Sotyktu® (deucravacitinib).
Sotyktu is a registered trademark of Bristol-Myers Squibb Company.
Results may vary.
Explore before-and-after photos
0 months
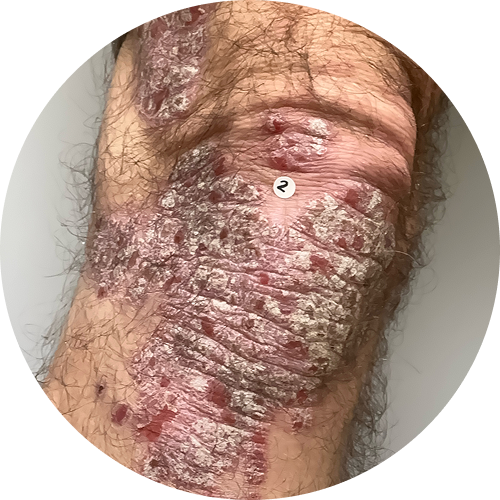
4 months (16 weeks)
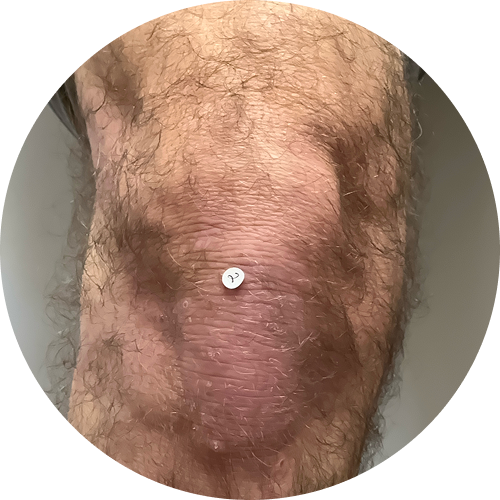
6 months (24 weeks)

0 months
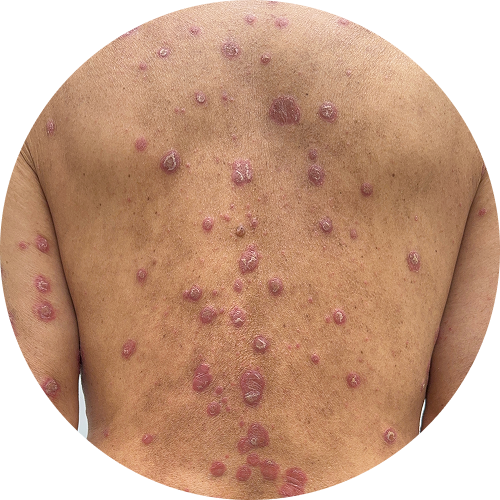
4 months (16 weeks)
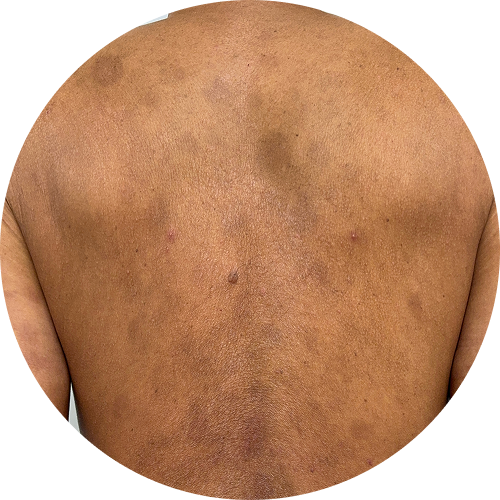
6 months (24 weeks)
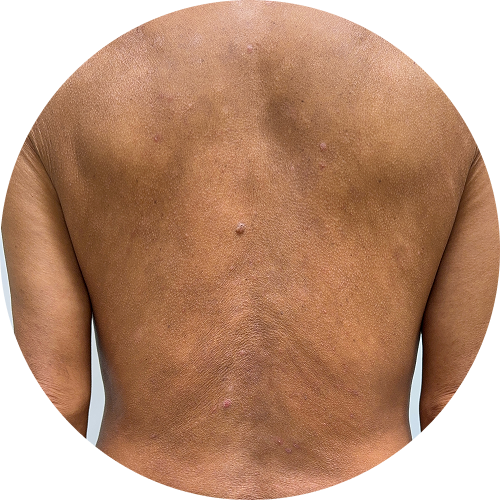
0 months
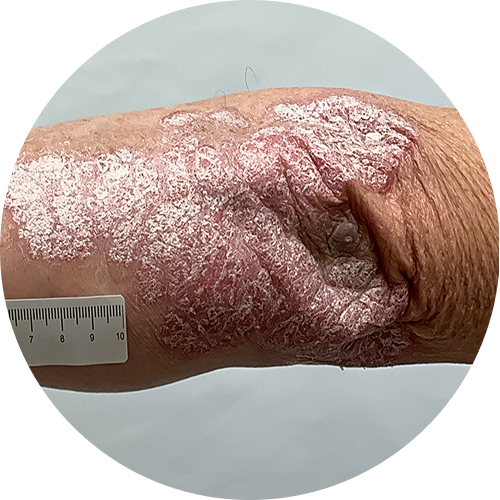
4 months (16 weeks)
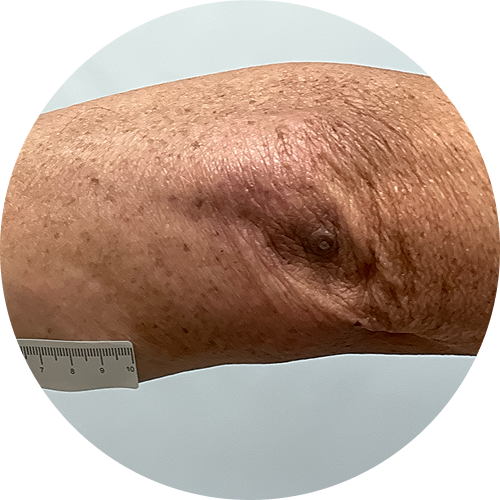
6 months (24 weeks)
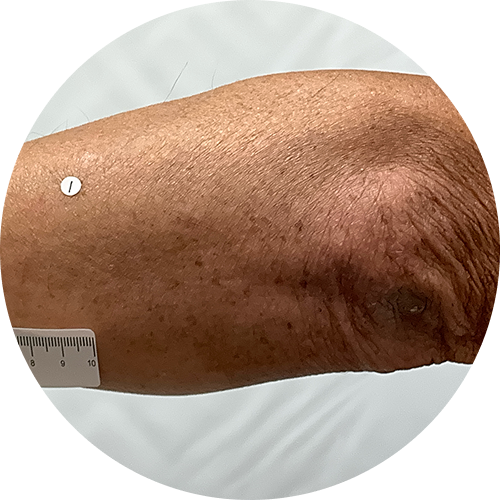
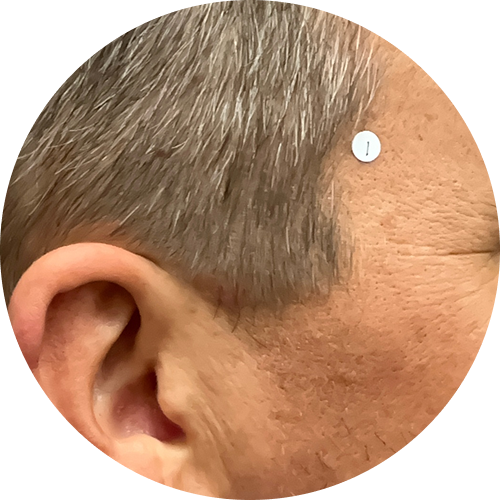
0 months
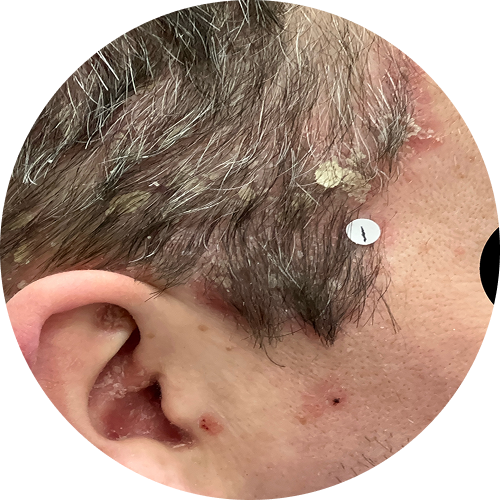
1 month (4 weeks)
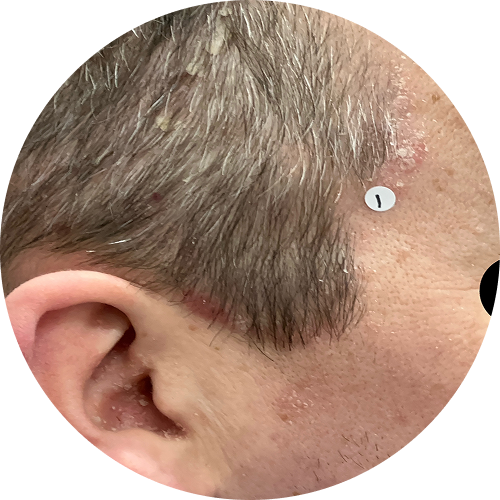
4 months (16 weeks)
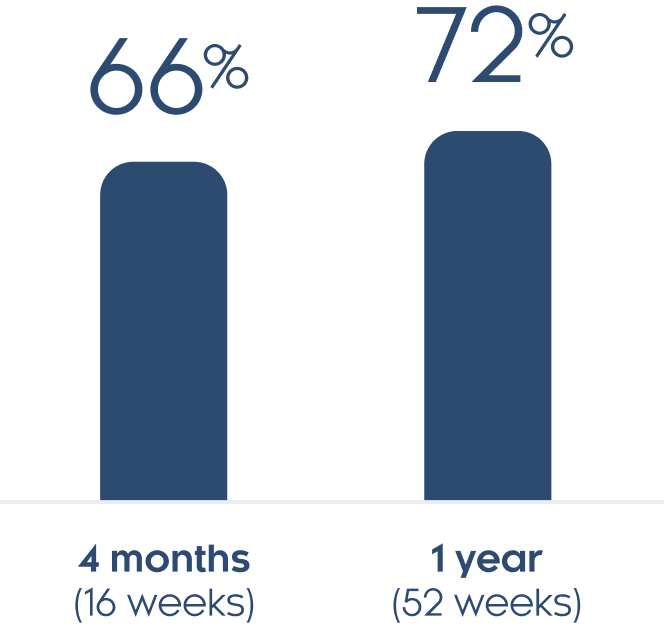
At 1 month (4 weeks) 15% of people with at least moderate scalp psoriasis had a clear or almost clear scalp. At 4 months (16 weeks) 66% of people with at least moderate scalp psoriasis had a clear or almost clear scalp.
Who was studied?
ICOTYDE was studied in 2 clinical trials, ICONIC-ADVANCE 1 and ICONIC-ADVANCE 2, including 1505 adults
with moderate to severe plaque psoriasis.
How was the study done?

People were randomly given
either a placebo, ICOTYDE, or
Sotyktu

Those who were given
a placebo
were switched to
ICOTYDE after
4 months (16 weeks)

Those who were given
SOTYKTU
were switched
to ICOTYDE after
6 months (24 weeks)
Patients were evaluated at different time points throughout the study. After 6 months (24 weeks), patients and healthcare providers knew that ICOTYDE was being used. This may have increased results.
A placebo is a pill with no medicine in it but looks the same and is taken the same way as the medicine being tested.
Who was studied?
ICOTYDE was studied in 2 clinical trials, ICONIC-ADVANCE 1 and ICONIC-ADVANCE 2, including 1505 adults
with moderate to severe plaque psoriasis.
How was the study done?

People were randomly given
either a placebo, ICOTYDE, or
Sotyktu

Those who were given
a placebo
were switched to
ICOTYDE after
4 months (16 weeks)

Those who were given
SOTYKTU
were switched
to ICOTYDE after
6 months (24 weeks)
Patients were evaluated at different time points throughout the study. After 6 months (24 weeks), patients and healthcare providers knew that ICOTYDE was being used. This may have increased results.
A placebo is a pill with no medicine in it but looks the same and is taken the same way as the medicine being tested.
Photos are of real patients. Images are owned by Janssen Biotech, Inc., a Johnson and Johnson company, from a clinical trial. Individual results may vary.
0 months

4 months (16 weeks)

6 months (24 weeks)

0 months

4 months (16 weeks)

6 months (24 weeks)

0 months

4 months (16 weeks)

6 months (24 weeks)

0 months

1 month (4 weeks)

4 months (16 weeks)

At 1 month (4 weeks) 15% of people with at least moderate scalp psoriasis had a clear or almost clear scalp. At 4 months (16 weeks) 66% of people with at least moderate scalp psoriasis had a clear or almost clear scalp.
In a clinical study of people with plaque psoriasis* with at least moderate severity in hard-to-treat areas
Most people (nearly 6 out of 10) saw clear or almost clear skin at 4 months (16 weeks)
After 4 months (16 weeks), patients and healthcare providers knew that ICOTYDE was being used. This may have increased results.
Scalp clearance
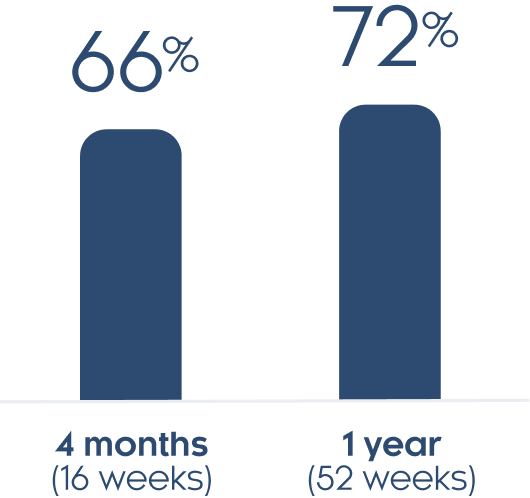
*In this study, people had plaque psoriasis on at least 1% of their body surface area.
Results may vary.
In a clinical study of people with plaque psoriasis* with at least moderate severity in hard-to-treat areas
Most people (nearly 6 out of 10) saw clear or almost clear skin at 4 months (16 weeks)
After 4 months (16 weeks), patients and healthcare providers knew that ICOTYDE was being used. This may have increased results.
Genital clearance


*In this study, people had plaque psoriasis on at least 1% of their body surface area.
Results may vary.
Who was studied?
ICOTYDE was studied in another clinical trial, ICONIC-TOTAL, including 311 people 12 and older with plaque psoriasis
affecting hard-to-treat areas with at least moderate severity like the scalp and genitals. In this study, people had plaque psoriasis on
at least 1% of their body surface area.
How was the study done?

Adolescents and adults were randomly
given either placebo or ICOTYDE

Those who were given a
placebo
were switched
to ICOTYDE after
4 months (16 weeks)
Patients were evaluated at different time points throughout the study. After 4 months (16 weeks), patients and healthcare providers knew that ICOTYDE was being used. This may have increased results.
A placebo is a pill with no medicine in it but looks the same and is taken the same way as the medicine being tested.

Who was studied?
ICOTYDE was studied in another clinical trial, ICONIC-TOTAL, including 311 people 12 and older with plaque psoriasis
affecting hard-to-treat areas with at least moderate severity like the scalp and genitals. In this study, people had plaque psoriasis on at least 1% of their body surface area.
How was the study done?
At start of study

Adolescents and adults were randomly given either placebo or ICOTYDE
4 months (16 weeks) into study

Those who were given a placebo
were switched
to ICOTYDE after
4 months (16 weeks)
Patients were evaluated at different time points throughout the study. After 4 months (16 weeks), patients and healthcare providers knew that ICOTYDE was being used. This may have increased results.
A placebo is a pill with no medicine in it but looks the same and is taken the same way as the medicine being tested.